
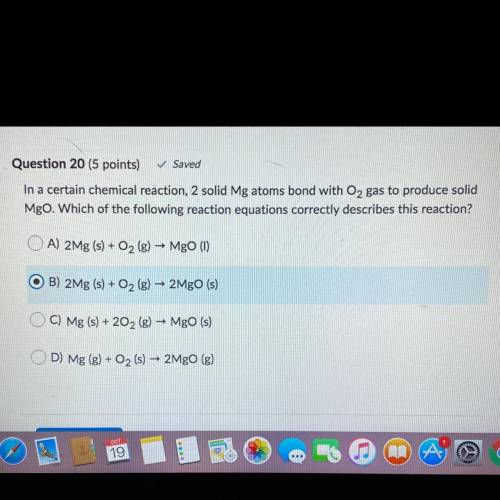
In a certain chemical reaction, 2 solid Mg atoms bond with O2 gas to produce solid
Mgo. Which of the following reaction equations correctly describes this reaction?
A) 2Mg (s) + O2(g) – Mgo (1)
O B) 2Mg (s) + O2 (g) – 2Mgo (s)
Sey
C) Mg (s) + 202 (g) – Mgo (s)
D) Mg (g) + O2 (s)
-
2MgO (g)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, gonzaleze18
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2


Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 23.06.2019 00:30, rose888829
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2
You know the right answer?
In a certain chemical reaction, 2 solid Mg atoms bond with O2 gas to produce solid
Mgo. Which of th...
Questions in other subjects:

Mathematics, 17.08.2021 16:10

English, 17.08.2021 16:10

Physics, 17.08.2021 16:10

Social Studies, 17.08.2021 16:10

Mathematics, 17.08.2021 16:10

Biology, 17.08.2021 16:10


Engineering, 17.08.2021 16:10




