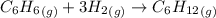The initial pressure of a mixture of C6H6 and an excess of H2 in a rigid vessel is 1.21 atm. A catalyst is introduced. After the reaction reaches completion, the temperature is restored to its initial value. The final pressure in the vessel is 0.839 atm. What was the mole fraction of C6H6 in the original mixture

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, robert7248
What is the cellular process that releases the energy stored in food molecules
Answers: 3

Chemistry, 22.06.2019 10:30, zayam1626
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 12:00, zamariahyou
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 12:30, skaterwolf1317
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2
You know the right answer?
The initial pressure of a mixture of C6H6 and an excess of H2 in a rigid vessel is 1.21 atm. A catal...
Questions in other subjects:

Mathematics, 24.10.2020 18:30


Mathematics, 24.10.2020 18:30

English, 24.10.2020 18:30

History, 24.10.2020 18:30

Arts, 24.10.2020 18:30

Mathematics, 24.10.2020 18:30

Mathematics, 24.10.2020 18:30

Mathematics, 24.10.2020 18:30