Chemistry, 16.10.2020 17:01 melissapulido198
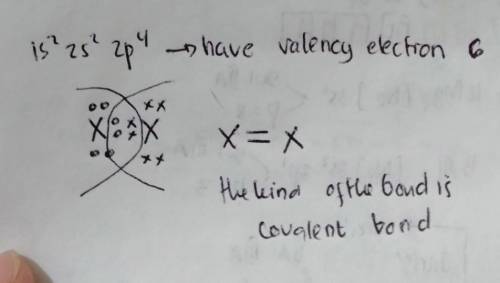
PLEASE HELP The electron configuration of an element is 1s 2s22p4. Describe what most likely happens when two atoms of this element move toward each other.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, jamesnaquan132
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 12:00, macylen3900
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2

You know the right answer?
PLEASE HELP
The electron configuration of an element is 1s 2s22p4. Describe what most likely happen...
Questions in other subjects:

Mathematics, 20.11.2021 06:40


Mathematics, 20.11.2021 06:40

Mathematics, 20.11.2021 06:40



Chemistry, 20.11.2021 06:40

Social Studies, 20.11.2021 06:40

History, 20.11.2021 06:40

History, 20.11.2021 06:40