
Chemistry, 16.10.2020 15:01 JvGaming2001
Calcium reacts with sulfur forming calcium sulfide. What is the theoretical yield (g) of CaS(s) that could be prepared from 7.19 g of Ca(s) and 2.67 g of sulfur(s)? Enter your answer with two decimal places. Do not type units with your answer.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, veronica25681
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 13:00, nadikadiaz1
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1


Chemistry, 23.06.2019 02:10, sativataurus
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2
You know the right answer?
Calcium reacts with sulfur forming calcium sulfide. What is the theoretical yield (g) of CaS(s) that...
Questions in other subjects:





Mathematics, 11.02.2022 14:00


Chemistry, 11.02.2022 14:00



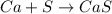
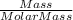
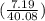 mol of Ca or 0.179 mol of Ca
mol of Ca or 0.179 mol of Ca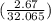 mol of S or 0.0833 mol of S
mol of S or 0.0833 mol of S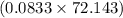 g or 6.01 g
g or 6.01 g

