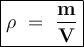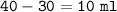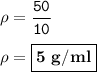Chemistry, 16.10.2020 06:01 daltonbrown4
Calculate the density of an object using the following information: Mass = 50 grams and the initial water level is 30-ml in a graduated cylinder, but rose to 40-ml after the object was dropped into it.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

You know the right answer?
Calculate the density of an object using the following information: Mass = 50 grams and the initial...
Questions in other subjects:

Social Studies, 11.11.2020 01:10

English, 11.11.2020 01:10


Mathematics, 11.11.2020 01:10


History, 11.11.2020 01:10

Physics, 11.11.2020 01:10

History, 11.11.2020 01:10

Mathematics, 11.11.2020 01:10

Mathematics, 11.11.2020 01:10