
Chemistry, 15.10.2020 18:01 catuchaljean1623
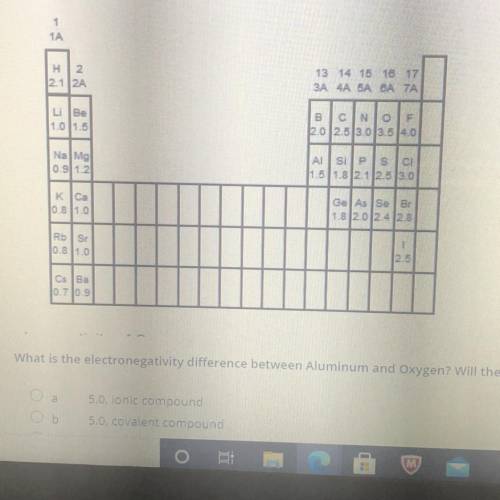
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. Ionic bonds generally form when the bonded elements have a difference in electronegativity greater than 1.5 What is the electronegativity difference between aluminum and oxygen will these two atoms form an iconic or covalent compound
A. 5.0 ionic compound
B. 5.0, covalent compound
C. 2.0, Covalent compound
D. 2.0, ionic compound

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, RedDemon59
Apeak with a retention time of 407 s has a width at half-height (w1/2) of 7.6 s. a neighboring peak is eluted 17 s later with a w1/2 of 9.4 s. a compound that is known not to be retained was eluted in 2.5 s. the peaks are not baseline resolved. how many theoretical plates would be needed to achieve a resolution of 1.5?
Answers: 2

Chemistry, 22.06.2019 18:00, jalenclarke25
What volume would 2.25 moles of ne has occupy at stp?
Answers: 1

Chemistry, 22.06.2019 21:00, rhondafits9000
Which property of water causes water drops to bead on a freshly waxed car?
Answers: 2

Chemistry, 23.06.2019 00:20, cmflores3245
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
Covalent bonds generally form when the bonded elements have a difference in electronegativity less t...
Questions in other subjects:

History, 21.12.2019 18:31

Mathematics, 21.12.2019 18:31



History, 21.12.2019 18:31

History, 21.12.2019 18:31


English, 21.12.2019 18:31


Biology, 21.12.2019 18:31



