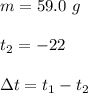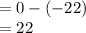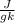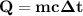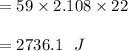At 1 atm, how much energy is required to heat 59.0 g H2O(s) at −22.0 ∘C to H2O(g) at 141.0 ∘C? STRATEGY: Calculate the energy needed for each temperature change or phase change individually. a. The energy needed to heat 59.0 g H2O(s) from −22.0 ∘C to its melting point. b. The energy needed to melt 59.0 g H2O(s) at its melting point. c. The energy needed to heat 59.0 g H2O(l) from the melting point to the boiling point. d. The energy needed to boil 59.0 g H2O(l) at its boiling point. e. The energy needed to heat 59.0 g H2O(g) from the boiling point to 141.0 ∘C. Sum the energies from each step and convert to kilojoules. Step 1. a. Going from −22.0 ∘C to the melting point of H2O (0 ∘C) is a temperature change of 22.0 ∘C. How much energy is needed to heat 59.0 g H2O(s) by 22.0 ∘C if its specific heat is 2.087 J/(g× ∘C)?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:50, Mercedes12152002
Where are chemicals found at work? a. only in cleaning products b. only in carpets and paint c. in every area of work d. only in food preparation submit
Answers: 1

Chemistry, 22.06.2019 00:30, megaaan214p61pb7
Which compounds have the empirical formula ch2o? a. c2h4o2 b. c3h6o3 c. ch2o2 d. c5h10o5 e. c6h12o6
Answers: 3

You know the right answer?
At 1 atm, how much energy is required to heat 59.0 g H2O(s) at −22.0 ∘C to H2O(g) at 141.0 ∘C? STRAT...
Questions in other subjects:

History, 14.10.2020 14:01


Mathematics, 14.10.2020 14:01


Mathematics, 14.10.2020 14:01


Mathematics, 14.10.2020 14:01

Engineering, 14.10.2020 14:01


Biology, 14.10.2020 14:01