check all that apply.

Why are most organic compounds nonconducting and insoluble in water?
check all that apply.
most organic compounds don't dissolve in water because they are polar.
most organic compounds don't dissolve in water because they are nonpolar.
they don't conduct electricity because they are ionic, not covalent.
they don't conduct electricity because they are covalent, not ionic.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, jusicca1109
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 13:30, Sbeech7246
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2
You know the right answer?
Why are most organic compounds nonconducting and insoluble in water?
check all that apply.
check all that apply.
Questions in other subjects:


Physics, 30.11.2020 20:10

English, 30.11.2020 20:10

History, 30.11.2020 20:10


Mathematics, 30.11.2020 20:10


Mathematics, 30.11.2020 20:10

Mathematics, 30.11.2020 20:10

English, 30.11.2020 20:10

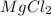 etc that are also polar in nature.
etc that are also polar in nature.


