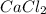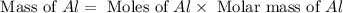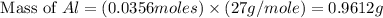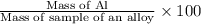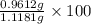5. A 1.1181-g sample of an alloy (a mixture) of aluminum and magnesium was treated with an
excess of sodium hydroxide solution. In the reaction, only the aluminum reacts with the sodium
hydroxide solution:
2 Al + 2 NaOH + 6 H202 Na[Al(OH)4] + 3 H2
If 0.1068 g of H2 is produced, what is the mass percent of aluminum in the alloy?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, angeljohnson2081
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1

Chemistry, 22.06.2019 13:30, richardwalker8ourhg2
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a. the mitochondria b. the nucleus c. the vacuoles d. the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 15:30, ricardotavarez6
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 19:30, Karinaccccc
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1
You know the right answer?
5. A 1.1181-g sample of an alloy (a mixture) of aluminum and magnesium was treated with an
excess o...
Questions in other subjects:

English, 01.02.2020 04:43



Mathematics, 01.02.2020 04:43


Mathematics, 01.02.2020 04:43


Mathematics, 01.02.2020 04:43

Biology, 01.02.2020 04:43

Mathematics, 01.02.2020 04:43

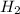 = 0.1068 g
= 0.1068 g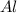 = 27 g/mol
= 27 g/mol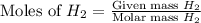
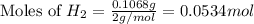
![2Al+2NaOH+6H_2O\rightarrow 2Na[Al(OH)_4]+3H_2](/tpl/images/0796/4861/bae6d.png)
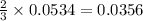 mole of
mole of