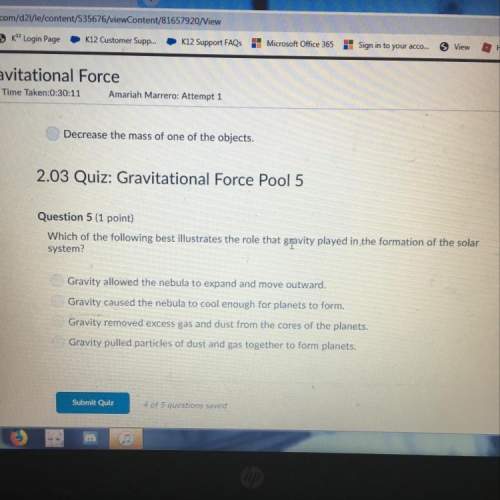
Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 17:10, codeyhatch142
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1


Chemistry, 22.06.2019 10:10, veronica022
Stage in which a typical star has completely stopped fusion
Answers: 1
You know the right answer?
On an x-y graph, where is the dependent variable scale (the marked increments)?...
Questions in other subjects:


Social Studies, 22.08.2019 07:30

History, 22.08.2019 07:30

Biology, 22.08.2019 07:30

History, 22.08.2019 07:30





Mathematics, 22.08.2019 07:30