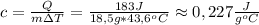Chemistry, 09.10.2019 19:00 Kykebailey2356
Calculate the specific heat (j/g∘c) for a 18.5-g sample of tin that absorbs 183 j when temperature increases from 35.0 ∘c to 78.6 ∘c.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1


Chemistry, 22.06.2019 15:30, lizzyhearts
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3
You know the right answer?
Calculate the specific heat (j/g∘c) for a 18.5-g sample of tin that absorbs 183 j when temperature i...
Questions in other subjects:


English, 08.03.2021 21:40

Mathematics, 08.03.2021 21:40




Mathematics, 08.03.2021 21:40