
Chemistry, 29.09.2020 14:01 andreyvaught2754
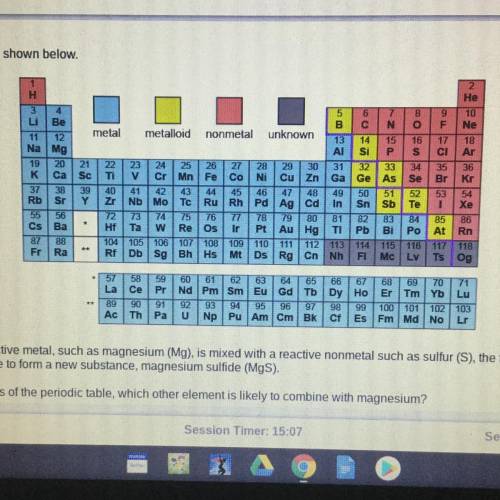
When a highly reactive metal, such as magnesium (Mg), is mixed with a reactive nonmetal such as sulfur (S), the two elements will
most likely combine to form a new substance, magnesium sulfide (MgS).
Based on the trends of the periodic table, which other element is likely to combine with magnesium?
A. Sodium (Na)
B. Helium (He)
C. Oxygen (O)
D. Gold (Au)

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:50, cj31150631
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 22.06.2019 20:00, Chynadoll94
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1
You know the right answer?
When a highly reactive metal, such as magnesium (Mg), is mixed with a reactive nonmetal such as sulf...
Questions in other subjects:




Biology, 24.06.2021 14:00

Business, 24.06.2021 14:00

Advanced Placement (AP), 24.06.2021 14:00

Biology, 24.06.2021 14:00

English, 24.06.2021 14:00


Mathematics, 24.06.2021 14:00



