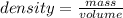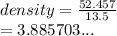Chemistry, 29.09.2020 04:01 dontcareanyonemo
Calculate the density of a material that has a mass of 52.457 g and a volume of 13.5 cm^3. The formula is D=m/v

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
You know the right answer?
Calculate the density of a material that has a mass of 52.457 g and a volume of 13.5 cm^3. The formu...
Questions in other subjects:


History, 10.07.2019 10:10

Social Studies, 10.07.2019 10:10


Social Studies, 10.07.2019 10:10



Business, 10.07.2019 10:10

Mathematics, 10.07.2019 10:10