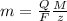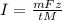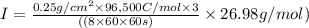The weight loss of an aluminum (Al) alloy corroding in HCI acid was observed to be 0.250 g/cm2 after an 8 h immersion period. What is the corresponding corrosion current density in mA/em2, assuming that all the corrosion is due to the reaction:
Al → Al3+ + 3e
The atomic weight of Al is 26.98 g/mol.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, batoolishak7475
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 23.06.2019 02:00, bagofmud8339
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1

Chemistry, 23.06.2019 15:30, shybunny7568
Sodium chloride can be made as follows: 2na + cl2 ? 2nacl i calculate the maximum amount of nacl possible if 2.3 g of sodium was reacted with excess chlorine. show all your workings.
Answers: 3
You know the right answer?
The weight loss of an aluminum (Al) alloy corroding in HCI acid was observed to be 0.250 g/cm2 after...
Questions in other subjects:



Business, 04.06.2020 18:59

Chemistry, 04.06.2020 18:59



English, 04.06.2020 18:59


Mathematics, 04.06.2020 18:59

Mathematics, 04.06.2020 18:59