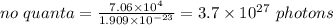Chemistry, 21.09.2020 09:01 mrmendrala
Heating 235 g of water from 22.6°C to 94.4°C in a microwave oven requires 7.06 × 104 J of energy. If the microwave frequency is 2.88 × 1010 s−1, how many quanta are required to supply the 7.06 × 104 J? The value for Planck's constant is 6.63 × 10-34 Jᐧs/quantum. The formula to use is Energy / (Planck's constant x Frequency).

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, pennygillbert
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 07:30, zamirareece17
1. list three scientific reasons cockroaches may fly.
Answers: 1


Chemistry, 22.06.2019 11:00, snowprincess99447
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
Heating 235 g of water from 22.6°C to 94.4°C in a microwave oven requires 7.06 × 104 J of energy. If...
Questions in other subjects:

Mathematics, 29.01.2021 22:00


English, 29.01.2021 22:00


Mathematics, 29.01.2021 22:00


Mathematics, 29.01.2021 22:00

Mathematics, 29.01.2021 22:00


Biology, 29.01.2021 22:00