
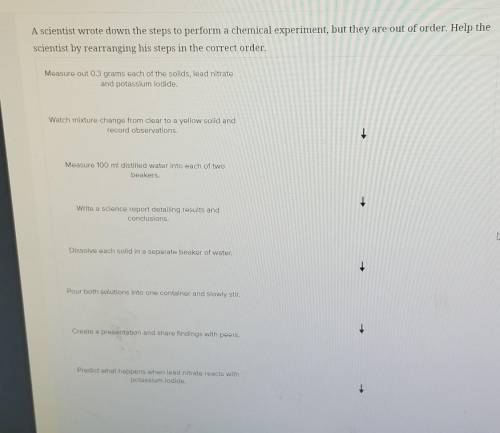
A scientist wrote down the steps to perform a chemical experiment, but they are out of order. Help the scientist by rearranging his steps in the correct order. Measure out 0.3 grams each of the solids, lead nitrate and potassium iodide. Watch mixture change from clear to a yellow solid and record observations. 1 Measure 100 ml distilled water into each of two beakers. 1 Write a science report detailing results and conclusions. Dissolve each solid in a separate beaker of water. Pour both solutions into one container and slowly stir. Create a presentation and share findings with peers. Predict what happens when lead nitrate reacts with potassium iodide.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, psychocatgirl1
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone, due to its high light availability and warm water temperature
Answers: 3


Chemistry, 22.06.2019 19:50, jakaylathomas11
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
A scientist wrote down the steps to perform a chemical experiment, but they are out of order. Help t...
Questions in other subjects:




Business, 14.01.2021 07:50



Physics, 14.01.2021 07:50

Mathematics, 14.01.2021 07:50

English, 14.01.2021 07:50



