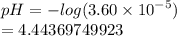Chemistry, 20.09.2020 05:01 thepantsgirl
Paul determines that the hydrogen ion concentration of his unknown solution is 3.60×10^-5 M. what is the pH of this solution?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:00, luisaareli6298
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 22.06.2019 18:20, juansebas35
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
Paul determines that the hydrogen ion concentration of his unknown solution is 3.60×10^-5 M. what is...
Questions in other subjects:

Mathematics, 17.02.2021 14:10

Computers and Technology, 17.02.2021 14:10

Biology, 17.02.2021 14:10

Geography, 17.02.2021 14:10




Biology, 17.02.2021 14:10

English, 17.02.2021 14:10

Chemistry, 17.02.2021 14:10

![p H = - log[ H^{ + } ]](/tpl/images/0768/5800/e313c.png)