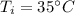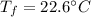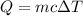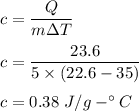Chemistry, 20.09.2020 05:01 jaymoney0531
If 5.0 g of copper cools from 35.0oC to 22.6 oC and loses 23.6 J of heat, what is the specific heat of copper? a. 0.038 J/(g. oC) b. 0.62 J/(g. oC) c. 0.076 J/(g. oC) d. 0.38 J/(g. oC)

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 11:30, samantha9430
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
If 5.0 g of copper cools from 35.0oC to 22.6 oC and loses 23.6 J of heat, what is the specific heat...
Questions in other subjects:





Spanish, 04.07.2019 13:30