
Chemistry, 06.09.2020 02:01 anthonybowie99
Calculate the mass of each of the following:
a. A sphere of gold with a radius of 10.5 cm. (The volume of a sphere with a radius r is V = (4/3)πr3; the density of gold is 19.3 g/cm^3.)
b. A cube of platinum of edge length 0.021 mm (density = 21.4 g/cm3).
c. 37.3 mL of ethanol (density = 0.798 g/mL).

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, fbillinton
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1



Chemistry, 22.06.2019 23:00, ceejay8005
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
Calculate the mass of each of the following:
a. A sphere of gold with a radius of 10.5 cm. (The vol...
Questions in other subjects:




Spanish, 09.11.2021 20:30




Mathematics, 09.11.2021 20:30

English, 09.11.2021 20:30

Mathematics, 09.11.2021 20:30

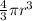
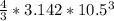 = 4849.68 cm^3
= 4849.68 cm^3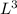 =
= 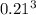 = 9.26 x 10^9 cm^3
= 9.26 x 10^9 cm^3

