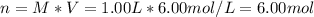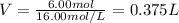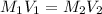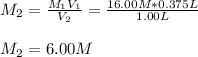Chemistry, 02.09.2020 04:01 matt199296
It is desired to make 1.00 liter of 6.00 M nitric acid from concentrated 16.00 M HNO3.A) How many moles of nitric acid are in 1.00 L of 6.00 M nitric acid?B) What volume of concentrated 16.00 M nitric acid will contain this number of moles?C) If this volume of concentrated nitric acid (answer to b) is diluted to 1.00 liter, what will be the molarity of the solution?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 17:40, aguilarjose
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 23.06.2019 02:30, elyzarobertson
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
It is desired to make 1.00 liter of 6.00 M nitric acid from concentrated 16.00 M HNO3.A) How many mo...
Questions in other subjects:






Spanish, 26.10.2021 05:20


Biology, 26.10.2021 05:20


History, 26.10.2021 05:20