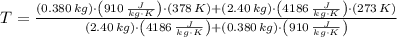Chemistry, 01.09.2020 02:01 davidcortez27
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then placed in 2.40 kg of water that is at 293 K. If the system is left to reach its equilibrium state, what will the equilibrium temperature be? Assume that no thermal energy is lost to the external environment. Use 4.186 J/(g x K) as the specific heat of water.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:30, sbush1412
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u. s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 17:00, davisnaziyahovz5sk
The arrangement of particles is most ordered in a sample of
Answers: 1


Chemistry, 23.06.2019 05:00, rosezgomez97
Asolution is made by dissolving 2.3 moles of sodium chloride (nacl) in 0.155 kilograms of water. if the molal boiling point constant for water (kb) is 0.51 °c/m, what would be the boiling point of this solution? show all the steps taken to solve this problem.
Answers: 1
You know the right answer?
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then...
Questions in other subjects:

History, 19.11.2020 15:50


Social Studies, 19.11.2020 15:50

Arts, 19.11.2020 15:50

Chemistry, 19.11.2020 15:50

Mathematics, 19.11.2020 15:50

Computers and Technology, 19.11.2020 15:50

Mathematics, 19.11.2020 15:50

Mathematics, 19.11.2020 15:50

Mathematics, 19.11.2020 15:50

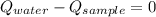
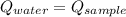
 - Heat received by water, measured in joules.
- Heat received by water, measured in joules. - Heat released by the sample of aluminium, measured in joules.
- Heat released by the sample of aluminium, measured in joules.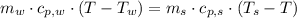
 ,
,  - Mass of water and the sample of aluminium, measured in kilograms.
- Mass of water and the sample of aluminium, measured in kilograms. ,
, 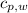 - Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.
- Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.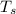 ,
,  - Initial temperatures of the sample of aluminium and water, measured in Kelvin.
- Initial temperatures of the sample of aluminium and water, measured in Kelvin. - Temperature which system reaches thermal equilibrium, measured in Kelvin.
- Temperature which system reaches thermal equilibrium, measured in Kelvin.

 ,
, 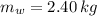 ,
,  ,
, 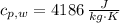 ,
, 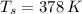 and
and  , the final temperature of the system is:
, the final temperature of the system is: