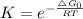Chemistry, 28.08.2020 16:01 j1theking18
Choose the best answer below. Which of the following reactions will have the largest equilibrium constant at 298 K?
a) 302(g) → 203(9) AGOrxn = +326 kJ
b) Mg(s) + N20(g) → Mgo(s) + N2(g) AG9rxn = -673.0 kJ
c) 2Hg(g) + O2(g) → 2HgO(s) AGºrx = -180.8 kJ
d) CaCO3(s) » Cao(s) + CO2(g) AG = +131.1 kJ
It is not possible to determine the reaction with the largest equilibrium constant using the given information.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, lindseyklewis1p56uvi
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1


Chemistry, 22.06.2019 15:30, elizabethprasad2
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 23.06.2019 00:00, chloe8979
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1
You know the right answer?
Choose the best answer below. Which of the following reactions will have the largest equilibrium con...
Questions in other subjects:



Physics, 23.04.2020 22:41


Mathematics, 23.04.2020 22:41

Mathematics, 23.04.2020 22:41



Social Studies, 23.04.2020 22:41