Chemistry, 25.08.2020 23:01 adaneri1234
g What is the half-life for a particular reaction if the rate law is rate = (1291 M⁻¹*min⁻¹)[A]² and the initial concentration of A is 0.250 M?

Answers: 2


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 14:30, Hannahmiller3773
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
You know the right answer?
g What is the half-life for a particular reaction if the rate law is rate = (1291 M⁻¹*min⁻¹)[A]² and...
Questions in other subjects:

Mathematics, 03.02.2020 18:56




Advanced Placement (AP), 03.02.2020 18:56


Arts, 03.02.2020 18:56

English, 03.02.2020 18:56


English, 03.02.2020 18:57

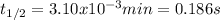
![t_{1/2}=\frac{1}{k[A]_0}](/tpl/images/0729/2407/6693f.png)