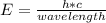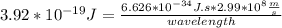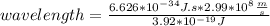Chemistry, 20.08.2020 01:01 mscharris66
The equation for photon energy, E, is E=hc/λ
where h = 6.626×10−34 J⋅s (Planck's constant) and c = 2.99×108 m/s (the speed of light).
What is the wavelength, λ, of a photon that has an energy of E = 3.92×10−19 J ?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, bakoeboo
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 21.06.2019 22:00, applereams
If a plot weight (in g) vs. volume (in ml) for a metal gave the equation y= 13.41x and r^2=0.9981 what is the density of the metal?
Answers: 2

Chemistry, 22.06.2019 09:10, chloeholt123
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
The equation for photon energy, E, is E=hc/λ
where h = 6.626×10−34 J⋅s (Planck's constant) and c =...
Questions in other subjects:


Physics, 27.06.2021 14:00



Biology, 27.06.2021 14:00

Social Studies, 27.06.2021 14:00


English, 27.06.2021 14:00

English, 27.06.2021 14:00