

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, KKHeffner02
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1


Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Calculate the [Cu2') remaining in 425 mL of a solution that was originally 0.366 M CuSO4 after passa...
Questions in other subjects:




Mathematics, 17.07.2020 20:01

English, 17.07.2020 20:01

Arts, 17.07.2020 20:01




Business, 17.07.2020 20:01

![[Cu]^{remaining}=0.357 M](/tpl/images/0724/2057/9ed71.png)
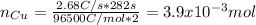

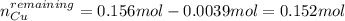
![[Cu]^{remaining}=0.152mol/0.425L](/tpl/images/0724/2057/98d78.png)


