Chemistry, 17.08.2020 01:01 cancerbaby209
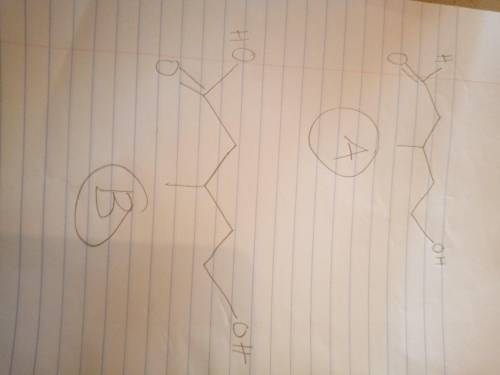
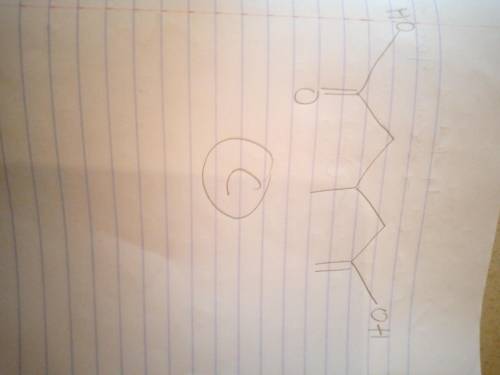
Compound A, C6H12O2, was found to be optically active, and it was slowly oxidized to an optically active carboxylic acid B, C6H12O3, by Ag(NH3)2. Oxidation of A by anhydrous CrO3 gave an optically inactive compound D that reacted with Zn amalgam/HCl to give 3-methylpentane. With aqueous H2CrO4, compound A was oxidized to an optically inactive dicarboxylic acid C, C6H10O4. Give structures for compounds A, B, and C; do not specify stereochemistry.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
Compound A, C6H12O2, was found to be optically active, and it was slowly oxidized to an optically ac...
Questions in other subjects:




English, 10.11.2020 23:00



Mathematics, 10.11.2020 23:00


Mathematics, 10.11.2020 23:00