Chemistry, 15.08.2020 22:01 raieliz1414
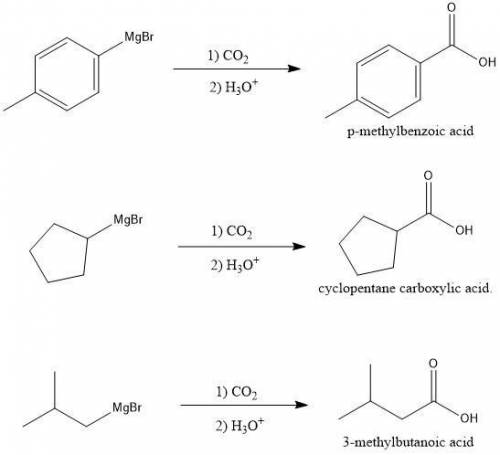
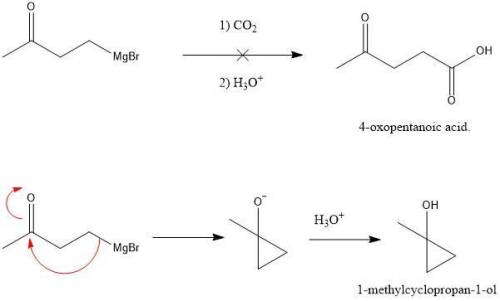
Which acid could not be prepared by treating a Grignard reagent with CO2?
a. p-methylbenzoic acid.
b. cyclopentane carboxylic acid.
c. 4-oxopentanoic acid.
d. 3-methylbutanoic acid.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:50, bellojamilet410
What substance has a mass of 9.5g and volume of 2.1cm^3
Answers: 2

Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 22.06.2019 23:40, sydneykated
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
You know the right answer?
Which acid could not be prepared by treating a Grignard reagent with CO2?
a. p-methylbenzoic acid....
Questions in other subjects:


Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01


Mathematics, 06.10.2020 14:01