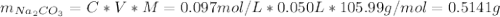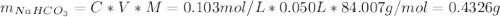Using only sodium carbonate, Na2CO3, sodium bicarbonate, NaHCO3, and distilled water determine how you could prepare 50.0 mL of a 0.20 M solution that is buffered to a pH of 10.3. The total molarity of the ions should be 0.20 M. The Ka of the hydrogen carbonate ion, HCO3 - , is 4.7 x 10-11 .

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2

Chemistry, 22.06.2019 17:30, TheViperMlg23676
What causes most sediment to wash or fall into a river
Answers: 1

Chemistry, 23.06.2019 07:30, apalacios3503
To separate a mixture of hard candies nd marbles the most efficient method would be
Answers: 3
You know the right answer?
Using only sodium carbonate, Na2CO3, sodium bicarbonate, NaHCO3, and distilled water determine how y...
Questions in other subjects:



Mathematics, 14.10.2019 23:30


Biology, 14.10.2019 23:30




Mathematics, 14.10.2019 23:30

![pH = pKa + log(\frac{[Na_{2}CO_{3}]}{[NaHCO_{3}]})](/tpl/images/0721/9986/3fe43.png)
![10.3 = -log(4.7 \cdot 10^{-11}) + log(\frac{[Na_{2}CO_{3}]}{[NaHCO_{3}]})](/tpl/images/0721/9986/394a4.png)
![\frac{[Na_{2}CO_{3}]}{[NaHCO_{3}]} = 0.94](/tpl/images/0721/9986/3fc8d.png) (1)
(1)![[Na_{2}CO_{3}] + [NaHCO_{3}] = 0.20 M](/tpl/images/0721/9986/3f8f2.png) (2)
(2)![[Na_{2}CO_{3}] = 0.20 - [NaHCO_{3}]](/tpl/images/0721/9986/5735e.png) (3)
(3) ![\frac{0.20 - [NaHCO_{3}]}{[NaHCO_{3}]} = 0.94](/tpl/images/0721/9986/bff59.png)
![0.94*[NaHCO_{3}] + [NaHCO_{3}] = 0.20](/tpl/images/0721/9986/5b36c.png)
![[NaHCO_{3}] = 0.103 M](/tpl/images/0721/9986/c3c9c.png)
![[Na_{2}CO_{3}] = 0.20 - [NaHCO_{3}] = 0.20 M - 0.103 M = 0.097 M](/tpl/images/0721/9986/fa97c.png)