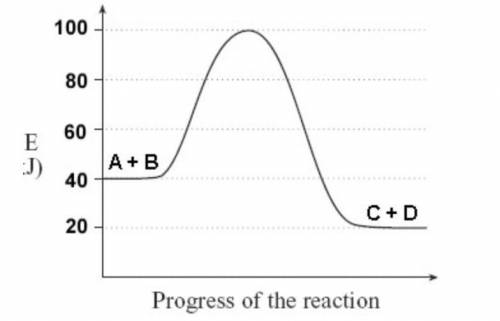
Using the following diagram, determine which of the statements below is true: The activation energy for the forward reaction is −60 J. The overall energy change for the forward reaction is −20 J. The activation energy for the reverse reaction is −80 J. The overall energy change for the reverse reaction is −40 J.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

You know the right answer?
Using the following diagram, determine which of the statements below is true: The activation energy...
Questions in other subjects:



Mathematics, 10.07.2019 04:30

Mathematics, 10.07.2019 04:30

Mathematics, 10.07.2019 04:30

Mathematics, 10.07.2019 04:30

History, 10.07.2019 04:30

French, 10.07.2019 04:30

Mathematics, 10.07.2019 04:30