Chemistry, 12.08.2020 18:01 Melymarrupe1345
Human lungs have evolved to breathe oxygen at a pressure as that in the atmosphere, 0.21 atm. If a particular heliox mixture to be carried by a scuba diver is at a pressure of 7.00 atm, what should be the partial pressure due to helium in order to maintain the pressure due to oxygen at 0.21 atm?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1

You know the right answer?
Human lungs have evolved to breathe oxygen at a pressure as that in the atmosphere, 0.21 atm. If a p...
Questions in other subjects:


History, 14.07.2019 16:30

Spanish, 14.07.2019 16:30

Mathematics, 14.07.2019 16:30


Health, 14.07.2019 16:30




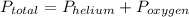 , where
, where  = total partial pressure of all the component gases in the mixture,
= total partial pressure of all the component gases in the mixture, 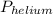 = partial pressure of helium gas, and
= partial pressure of helium gas, and 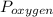 = partial pressure of oxygen gas.
= partial pressure of oxygen gas.