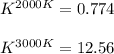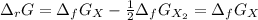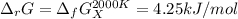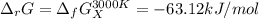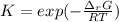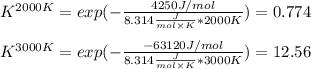Chemistry, 12.08.2020 04:01 anitadefrances
The equation represents the decomposition of a generic diatomic element in its standard state. 12X2(g)⟶X(g) Assume that the standard molar Gibbs energy of formation of X(g) is 4.25 kJ·mol−1 at 2000. K and −63.12 kJ·mol−1 at 3000. K. Determine the value of K (the thermodynamic equilibrium constant) at each temperature.

Answers: 1


Other questions on the subject: Chemistry



You know the right answer?
The equation represents the decomposition of a generic diatomic element in its standard state. 12X2(...
Questions in other subjects:






Biology, 02.10.2019 17:30

Mathematics, 02.10.2019 17:30


Advanced Placement (AP), 02.10.2019 17:30

Mathematics, 02.10.2019 17:30