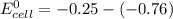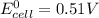Chemistry, 12.08.2020 04:01 kingjames82
Using these metal ion/metal standard reduction potentials Cd2+(aq)|Cd(s) Zn2+(aq)|Zn(s) Ni2+(aq)|Ni(s) Cu2+(aq)/Cu(s) -0.40 V -0.76 V ‑0.25 V +0.34 V Calculate the standard cell potential for the cell whose reaction is Ni2+(aq) + Zn(s) →Zn2+(aq)+ Ni(s)

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 18:00, darrell1168
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
Using these metal ion/metal standard reduction potentials Cd2+(aq)|Cd(s) Zn2+(aq)|Zn(s) Ni2+(aq)|Ni(...
Questions in other subjects:



Physics, 17.10.2021 03:20

Mathematics, 17.10.2021 03:20


Mathematics, 17.10.2021 03:20

Mathematics, 17.10.2021 03:20

Physics, 17.10.2021 03:20


Spanish, 17.10.2021 03:20

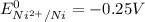
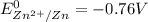
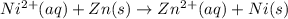
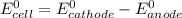
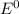 are standard reduction potentials.
are standard reduction potentials.