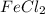Chemistry, 12.08.2020 05:01 tishfaco5000
Ytu perform an electrolysis reaction using FeCl, iron (1) chloride. Which of the following will happen?
A. Chlorine will move to the positive electrode and become reduced.
B. Iron will move to the positive electrode and become oxidized.
C. Chlorine will move to the negative electrode and become oxidized.
D. Iron will move to the negative electrode and become reduced.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, thebrain1345
Plz me get these answer dubble cheak ur answer plz ppl i need it right
Answers: 2

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
Ytu perform an electrolysis reaction using FeCl, iron (1) chloride. Which of the following will happ...
Questions in other subjects:

History, 22.12.2019 17:31


Social Studies, 22.12.2019 17:31


Chemistry, 22.12.2019 17:31




Social Studies, 22.12.2019 17:31

History, 22.12.2019 17:31