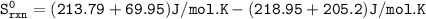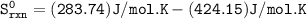Predict the sign and calculate ΔS° for a reaction. Close Problem Consider the reaction H2CO(g) + O2(g)CO2(g) + H2O(l) Based upon the stoichiometry of the reaction the sign of Sºrxn should be . Using standard thermodynamic data (in the Chemistry References), calculate Sºrxn at 25°C. Sºrxn = J/K•mol

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:30, toriabrocks
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2

Chemistry, 22.06.2019 20:20, catchonyet
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
You know the right answer?
Predict the sign and calculate ΔS° for a reaction. Close Problem Consider the reaction H2CO(g) + O2(...
Questions in other subjects:






Social Studies, 28.06.2019 00:40


Social Studies, 28.06.2019 00:40

Social Studies, 28.06.2019 00:40

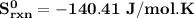
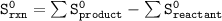
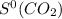 = 213.79 J/mol.K
= 213.79 J/mol.K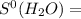 69.95 J/mol.K
69.95 J/mol.K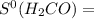 218.95 J/mol.K
218.95 J/mol.K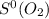 = 205.2 J/mol.K
= 205.2 J/mol.K