Chemistry, 12.08.2020 06:01 iamabeast51
A chemist fills a reaction vessel with 0.978 g aluminum hydroxide AlOH3 solid, 0.607 M aluminum Al+3 aqueous solution, and 0.396 M hydroxide OH− aqueous solution at a temperature of 25.0°C.
Under these conditions, calculate the reaction free energy ΔG for the following chemical reaction:
Al(OH)3(s) = A1+ (aq) +30H (aq)
Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule.
KJ

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, momof7hardings
When would a bouncy ball have the most potential energy
Answers: 2

Chemistry, 22.06.2019 06:00, rigobertogarza2
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

You know the right answer?
A chemist fills a reaction vessel with 0.978 g aluminum hydroxide AlOH3 solid, 0.607 M aluminum Al+3...
Questions in other subjects:



Mathematics, 31.03.2021 19:00


Mathematics, 31.03.2021 19:00


Mathematics, 31.03.2021 19:00

Mathematics, 31.03.2021 19:00


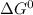 = 168.12 kJ
= 168.12 kJ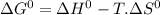
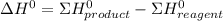
![\Delta H^{0} = [3*(-299.9)+(-524.7)] - (-1277)](/tpl/images/0719/2708/1f302.png)
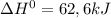
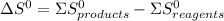
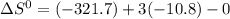
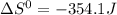
![\Delta G^{0} = 62600 - [298.(-354.1)]](/tpl/images/0719/2708/9ef3e.png)