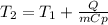Chemistry, 12.08.2020 07:01 FlowerChild1229
A 135 g sample of H20 at 85°C is cooled. The water loses a total of 15 kJ of energy in the cooling
process. What is the final temperature of the water? The specific heat of water is 4.184 J/g.°C.
A. 112°C
B. 58°C
C. 70°C
D. 84°C
E. 27°C

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, aydenmasonc
Which statement describes james chadwick’s discovery.
Answers: 2

Chemistry, 22.06.2019 13:50, aesthetickait
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
You know the right answer?
A 135 g sample of H20 at 85°C is cooled. The water loses a total of 15 kJ of energy in the cooling...
Questions in other subjects:

Mathematics, 09.04.2020 06:53

Mathematics, 09.04.2020 06:53

Physics, 09.04.2020 06:53

Mathematics, 09.04.2020 06:53


Mathematics, 09.04.2020 06:53

Social Studies, 09.04.2020 06:54


Mathematics, 09.04.2020 06:54

Biology, 09.04.2020 06:54

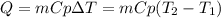
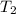 we obtain:
we obtain: