
Chemistry, 12.08.2020 06:01 munziruddin204
Consider the following: Li(s) + ½ I₂(g) --> LiI(s) ΔH = –292 kJ. LiI(s) has a lattice energy of –753 kJ/mol. The ionization energy of Li(g) is 520 kJ/mol, the bond energy of I₂(g) is 151 kJ/mol, and the electron affinity of I(g) is –295 kJ/mol. Use these data to determine the heat of sublimation of Li(s).

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, monithebtslover01
Find the empirical formula of each of the following compounds. given mass or for each element in a sample of the compound 3,611 g ca; 6.389 g c1
Answers: 1

Chemistry, 22.06.2019 05:30, greekfreekisdbz
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3


Chemistry, 22.06.2019 12:50, khorasanpublic
The number at the end of an isotope’s name is the number.
Answers: 1
You know the right answer?
Consider the following: Li(s) + ½ I₂(g) --> LiI(s) ΔH = –292 kJ. LiI(s) has a lattice energy of –...
Questions in other subjects:

History, 15.01.2020 10:31


History, 15.01.2020 10:31

Mathematics, 15.01.2020 10:31

Mathematics, 15.01.2020 10:31

Mathematics, 15.01.2020 10:31



Biology, 15.01.2020 10:31

English, 15.01.2020 10:31

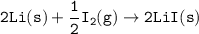
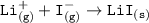
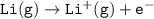
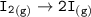
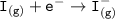
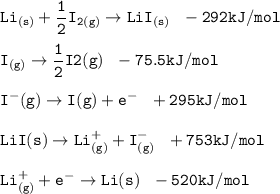
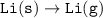 = (-292 +(-75.5)+295+753+(-520)) kJ/mol
= (-292 +(-75.5)+295+753+(-520)) kJ/mol

