
Chemistry, 04.08.2020 23:01 elijahjacksonrp6z2o7
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and 0.850 atm. Show your work. Part 2. If this sample was placed under extremely low temperature, describe how the actual volume would compare to the predicted volume. Explain your answer.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, Ezekielcassese
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 10:30, kylemartinez13
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2


Chemistry, 22.06.2019 17:30, Naysa150724
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3
You know the right answer?
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and...
Questions in other subjects:



Computers and Technology, 16.11.2019 01:31

Geography, 16.11.2019 01:31

Mathematics, 16.11.2019 01:31



Biology, 16.11.2019 01:31

Biology, 16.11.2019 01:31


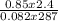
 = 0.087mole
= 0.087mole
 =7.15gmol⁻¹
=7.15gmol⁻¹


