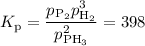Chemistry, 03.08.2020 14:01 KetaFord1978
When a mixture of PH3, P2, and H2 comes to equilibrium at 873 K, PP2= 0.422 atm and PH2= 0.832 atm. What is PPH3? Express your answer with the appropriate units.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
When a mixture of PH3, P2, and H2 comes to equilibrium at 873 K, PP2= 0.422 atm and PH2= 0.832 atm....
Questions in other subjects:


English, 14.01.2021 19:10



Mathematics, 14.01.2021 19:10




Mathematics, 14.01.2021 19:10

Law, 14.01.2021 19:10