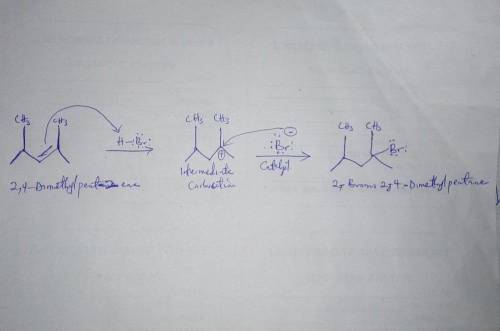
2,4-Dimethylpent-2-ene undergoes an electrophilic addition reaction in the presence of HBr to form 2-bromo-2,4-dimethylpentane. Complete the mechanism of this addition and draw the intermediates formed as the reaction proceeds. Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. Electron flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, georgehall3027
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 06:00, coolkid2041
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 09:30, jewelz5887
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1
You know the right answer?
2,4-Dimethylpent-2-ene undergoes an electrophilic addition reaction in the presence of HBr to form 2...
Questions in other subjects:

History, 21.09.2021 07:50

Biology, 21.09.2021 07:50





Mathematics, 21.09.2021 07:50

History, 21.09.2021 07:50

Mathematics, 21.09.2021 07:50