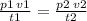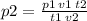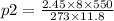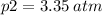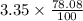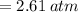Chemistry, 27.07.2020 23:01 lydia1melton
Air contains 78.08% nitrogen, 20.095% oxygen, and 0.93% argon. A sample of air occupies 8 L at 273 K and applies a pressure of 2.45 atm to its container. The sample of air is heated to 550 K, at which time it takes up 11.8 L. What is the partial pressure of nitrogen after the air has been heated? A. 2.61 atm B. 261.57 atm C. 282.16 atm D. 1.91 atm

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, madisonrosamond99
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 10:00, ellaemtagedeane
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 22.06.2019 16:50, TrueKing184
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1
You know the right answer?
Air contains 78.08% nitrogen, 20.095% oxygen, and 0.93% argon. A sample of air occupies 8 L at 273 K...
Questions in other subjects:

Mathematics, 14.03.2020 06:20


Mathematics, 14.03.2020 06:21

Health, 14.03.2020 06:21





Arts, 14.03.2020 06:23

Mathematics, 14.03.2020 06:23