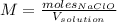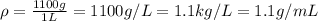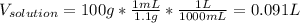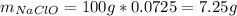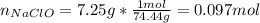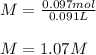Chemistry, 27.07.2020 01:01 santana647
1L of bleach has a mass of 1,100 grams, 7.25% of the mass of bleach is NaClO, 1 mol of NaClO has a mass of 74.44 grams. What is the molarity (mol/L) of NaClO in the bleach? A.0.097 B.0.93 C. 1.07 D.79.75

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, natannale
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1


Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

You know the right answer?
1L of bleach has a mass of 1,100 grams, 7.25% of the mass of bleach is NaClO, 1 mol of NaClO has a m...
Questions in other subjects:

History, 02.02.2021 01:30



Mathematics, 02.02.2021 01:30


History, 02.02.2021 01:30


Mathematics, 02.02.2021 01:30


Social Studies, 02.02.2021 01:30