

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:10, maribel2421
Starch and are common polysaccharide carbohydrates found in plants. sucrose glycogen fructose cellulose
Answers: 3

Chemistry, 22.06.2019 04:00, eborkins
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

You know the right answer?
Consider the reaction 2N2(g) O2(g)2N2O(g) Using the standard thermodynamic data in the tables linked...
Questions in other subjects:


Biology, 12.12.2019 08:31

History, 12.12.2019 08:31

Chemistry, 12.12.2019 08:31



Mathematics, 12.12.2019 08:31

Mathematics, 12.12.2019 08:31


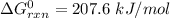

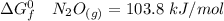
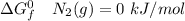

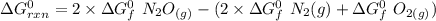
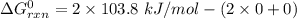
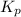 can be expressed as :
can be expressed as :
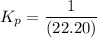

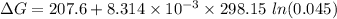
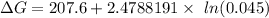
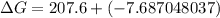
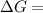 199.912952 kJ
199.912952 kJ

