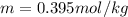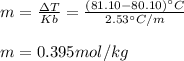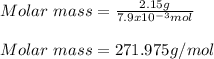Chemistry, 25.07.2020 06:01 AeelynRamos
Benzene boils at 80.10 °C and has a molal boiling constant, k b, of 2.53 C/m. When 2.15 g of a compound is dissolved in 20.0 g of benzene, the resulting solution has a boiling point of 81.10 °C. What is the molality of the solute?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, tahjaybenloss16
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 10:10, alvaradolm6853
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 23:10, RealStephani
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(s o4)2·7h2omgso4·7h2o
Answers: 1
You know the right answer?
Benzene boils at 80.10 °C and has a molal boiling constant, k b, of 2.53 C/m. When 2.15 g of a compo...
Questions in other subjects:

Mathematics, 30.08.2019 14:10

History, 30.08.2019 14:10

History, 30.08.2019 14:10

Mathematics, 30.08.2019 14:10


Mathematics, 30.08.2019 14:10


History, 30.08.2019 14:10


Biology, 30.08.2019 14:10