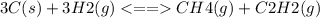Chemistry, 23.07.2020 06:01 quintencoffman2
Consider the following system, which is at equilibrium, 3C(s) + 3H2(g) <--> CH4(g) + C2H2(g) The result of removing some C(s) from the system will be:

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:50, shaylawaldo11
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 16:50, brandiwingard
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
Consider the following system, which is at equilibrium, 3C(s) + 3H2(g) <--> CH4(g) + C2H2(g) T...
Questions in other subjects:

Spanish, 19.11.2020 05:50


Spanish, 19.11.2020 05:50





Mathematics, 19.11.2020 05:50

Mathematics, 19.11.2020 05:50