
Chemistry, 23.07.2020 19:01 dustonangiecook
chromate is sparingly soluble in aqueous solutions. The Ksp of Ag2CrO4 is 1.12×10−12 . What is the solubility (in mol/L) of silver chromate in 1.00 M potassium chromate aqueous solution?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 21:50, donttrip10
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state. a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2


Chemistry, 23.06.2019 05:00, pmbeachy3102
If 15 drops of ethanol from a medicine dropper weigh 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? density of ethanol is ethanol is 0.80g/ml.
Answers: 2
You know the right answer?
chromate is sparingly soluble in aqueous solutions. The Ksp of Ag2CrO4 is 1.12×10−12 . What is the s...
Questions in other subjects:

Mathematics, 28.10.2020 02:30

English, 28.10.2020 02:30

English, 28.10.2020 02:30

Mathematics, 28.10.2020 02:30

Computers and Technology, 28.10.2020 02:30



Mathematics, 28.10.2020 02:30


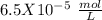
 , so:
, so: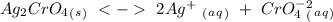
![Kps~=~[Ag^+]^2[CrO_4^-^2]](/tpl/images/0711/8854/b7f4a.png)
 formed, 2 moles of
formed, 2 moles of 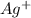 are formed. We can use "X" for the unknown concentration of each ion, so:
are formed. We can use "X" for the unknown concentration of each ion, so:![[CrO_4^-^2]~=~X](/tpl/images/0711/8854/4fd99.png) and
and ![[Ag^+]~=~2X](/tpl/images/0711/8854/da6f8.png)







