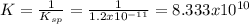Chemistry, 20.07.2020 01:01 littledudefromacross
If 75.0 mL of a 2.63 · 10-3 M NaOH is mixed with 125.0 mL of 1.80 · 10-3 M MgCl2, then calculate the reaction quotient and state if a precipitate will form? The Ksp of the expected precipitate is 1.2 · 10-11.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, zaehairston78531
What is the nature of the ca-cl bond in a molecule of calcium chloride (cacl2) if the electronegativity value of calcium is 1.0 and that of chlorine is 3.16?
Answers: 1


Chemistry, 22.06.2019 00:00, ashleyjaslin
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 23:40, tilievaughn14
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1
You know the right answer?
If 75.0 mL of a 2.63 · 10-3 M NaOH is mixed with 125.0 mL of 1.80 · 10-3 M MgCl2, then calculate the...
Questions in other subjects:


Geography, 05.04.2021 15:10


History, 05.04.2021 15:10


English, 05.04.2021 15:20

Social Studies, 05.04.2021 15:20

Social Studies, 05.04.2021 15:20

Mathematics, 05.04.2021 15:20

Mathematics, 05.04.2021 15:20

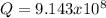

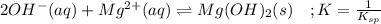
![\frac{1}{K_{sp}}=\frac{1}{[OH^-]^2[Mg^{2+}]}](/tpl/images/0709/8931/c7f27.png)
![[OH^-]=\frac{75.0mL*2.63x10^{-3}M}{75.0mL+125.0mL}=9.86x10^{-4}M](/tpl/images/0709/8931/2f439.png)
![[Mg^{2+}]=\frac{125.0mL*1.80x10^{-3}M}{75.0mL+125.0mL}=1.125x10^{-3}M](/tpl/images/0709/8931/0caa8.png)
![Q=\frac{1}{[OH^-]^2[Mg^{2+}]}=\frac{1}{(9.86x10^{-4})^2*1.125x10^{-3}} =9.143x10^8](/tpl/images/0709/8931/2e242.png)