
Chemistry, 19.07.2020 01:01 josephfoxworth
A solution contains 0.0440 M Ca2 and 0.0940 M Ag. If solid Na3PO4 is added to this mixture, which of the phosphate species would precipitate out of solution first?
A. Na3PO4.
B. Ag3PO4.
C. Ca3(PO4)2
When the second cation just starts to precipitate, what percentage of the first cation remains in solution?

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:30, psychocatgirl1
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone, due to its high light availability and warm water temperature
Answers: 3

You know the right answer?
A solution contains 0.0440 M Ca2 and 0.0940 M Ag. If solid Na3PO4 is added to this mixture, which of...
Questions in other subjects:

Biology, 17.07.2019 03:40

Mathematics, 17.07.2019 03:40


History, 17.07.2019 03:40




Mathematics, 17.07.2019 03:40

History, 17.07.2019 03:40

English, 17.07.2019 03:40

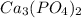 will precipitate out first
will precipitate out first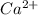 remaining = 12.86%
remaining = 12.86%![[Ca^{2+}] = 0.0440 \ M](/tpl/images/0709/5794/07981.png)
![[Ag^+] = 0.0940 \ M](/tpl/images/0709/5794/e77de.png)
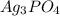
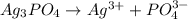
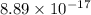
![Ksp = [Ag^+]^3[PO_4^{3-}]](/tpl/images/0709/5794/f170d.png)
![8.89 \times 10 ^{-17} = (0.0940)^3[PO_4^{3-}]](/tpl/images/0709/5794/f1b1a.png)
![\dfrac{8.89 \times 10 ^{-17}}{(0.0940)^3} = [PO_4^{3-}]](/tpl/images/0709/5794/fb0ea.png)
![[PO_4^{3-}] =\dfrac{8.89 \times 10 ^{-17}}{(0.0940)^3}](/tpl/images/0709/5794/36b7f.png)
![[PO_4^{3-}] =1.07 \times 10^{-13}](/tpl/images/0709/5794/d9e0c.png)
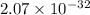
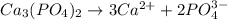
![Ksp = [Ca^{2+}]^3 [PO_4^{3-}]^2](/tpl/images/0709/5794/409cc.png)
![2.07 \times 10^{-33} = (0.0440)^3 [PO_4^{3-}]^2](/tpl/images/0709/5794/a627b.png)
![\dfrac{2.07 \times 10^{-33} }{(0.0440)^3}= [PO_4^{3-}]^2](/tpl/images/0709/5794/4ad8e.png)
![[PO_4^{3-}]^2 = \dfrac{2.07 \times 10^{-33} }{(0.0440)^3}](/tpl/images/0709/5794/fbe62.png)
![[PO_4^{3-}]^2 = 2.43 \times 10^{-29}](/tpl/images/0709/5794/43b90.png)
![[PO_4^{3-}] = \sqrt{2.43 \times 10^{-29}](/tpl/images/0709/5794/0afa0.png)
![[PO_4^{3-}] =4.93 \times 10^{-15}](/tpl/images/0709/5794/19717.png)
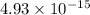 in
in 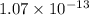
![[Ca^+]](/tpl/images/0709/5794/2b2af.png) when the second cation starts to precipitate ; we have :
when the second cation starts to precipitate ; we have :![2.07 \times 10^{-33} = [Ca^{2+}]^3 (1.07 \times 10^{-13})^2](/tpl/images/0709/5794/8e719.png)
![[Ca^{2+}]^3 = \dfrac{2.07 \times 10^{-33} }{(1.07 \times 10^{-13})^2}](/tpl/images/0709/5794/5de75.png)
![[Ca^{2+}]^3 =1.808 \times 10^{-7}](/tpl/images/0709/5794/9d3e5.png)
![[Ca^{2+}] =\sqrt[3]{1.808 \times 10^{-7}}](/tpl/images/0709/5794/21597.png)
![[Ca^{2+}] =0.00566](/tpl/images/0709/5794/67420.png)
![[Ca^{2+}]](/tpl/images/0709/5794/17576.png) in the solution is 0.00566
in the solution is 0.00566

