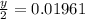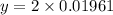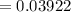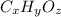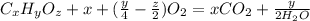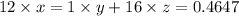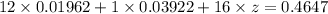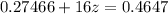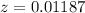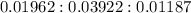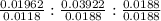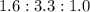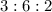Chemistry, 17.07.2020 23:01 ishrael2001
A 0.4647-g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxygen to yield 0.01962 mol of CO2 and 0.01961 mol of H2O. The empirical formula of the compound was found to be C3H6O2

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, shreyapatel2004
How many atoms of oxygen are contained in 160 grams of n2o3
Answers: 2

Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 07:30, tntaylor862
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 1

Chemistry, 22.06.2019 15:30, christopherluckey7
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
A 0.4647-g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxy...
Questions in other subjects:

Mathematics, 07.01.2021 03:50



English, 07.01.2021 03:50

Mathematics, 07.01.2021 03:50


Mathematics, 07.01.2021 03:50



Mathematics, 07.01.2021 03:50