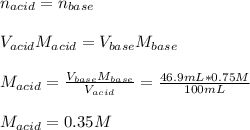Chemistry, 17.07.2020 20:01 mrashrafkotkaat
The reaction of perchloric acid (HClO4) with lithium hydroxide (LiOH) is described by the equation: HClO4 + LiOH → LiClO4 + H2O Suppose 100 mL of perchloric acid is neutralized by exactly 46.9 mL of 0.75 M lithium hydroxide. What is the concentration of the perchloric acid? A. 0.35 M B. 0.47 M C. 0.63 M D. 1.60 M

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, qwerty8364
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1


Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 17:00, abbygailgo674
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1
You know the right answer?
The reaction of perchloric acid (HClO4) with lithium hydroxide (LiOH) is described by the equation:...
Questions in other subjects:

Mathematics, 20.03.2020 04:00



Mathematics, 20.03.2020 04:01





Mathematics, 20.03.2020 04:01